The smooth green surface of a rock sits just below a pond’s surface. Its deceptively uniform green coating is teeming with life: algae, bacteria, and other microorganisms. They all depend on sunlight for energy and are constantly competing with each other for better access to light. While some microbes are immobile, stuck on to the rock in their original positions, others are mobile and move around on the rock’s surface in response to light. In the past, little was known about how bacteria perform phototaxis, motion in response to light. Recently however, researchers discovered that cyanobacteria, a subset of bacteria able to perform photosynthesis, act like tiny eyes, able to focus light internally and move towards its source.
Synechocystis, the species of cyanobacteria used in this study, is spherically shaped. As a result, it can focus light to the back side of the cell, so that the region of the cell farthest from the light source is the brightest. The bacteria respond to this brightness, moving away from that side and advancing towards the light. Synechocystis move using pili, hair like strands that coat the surface of the cell and attach to nearby surfaces, dragging the cell along in a jerking motion, in this case towards a light source.
This study shows that the mechanism of phototaxis is completely different than that of chemotaxis, cellular motion in response to a chemical stimulus. Cells that perform chemotaxis have long protrusions called flagella that rotate in liquid like propellers and direct the cell’s motion. They move towards or away from chemicals using a combination of continuous motion and tumbles, stops with random changes in direction. With each change in direction, the cell’s goal is to move towards a more desirable chemical concentration. When it reaches a beneficial concentration, the bacterium continues on its course, but once it becomes adjusted to the new concentration, tumbling returns.
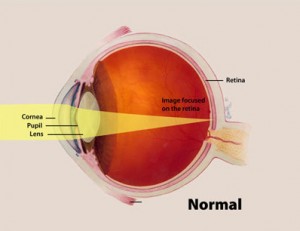
The cells detect light using a method similar to that of the human eye: the side of the cell receiving light acts like a convex lens, bringing all of the angles of a light source together into a focal point. Within a human eye, this focal point aligns with the retina, so the eye can communicate with the brain. In cyanobacteria, the focal point is at the back of the cell, where light is intensified and the pili shrink, moving the cell closer to the light source. This mechanism of direct detection is unprecedented. “This is something that was not actually known in bacteria before,” said Conrad Mullineaux, microbiology professor and a lead scientist on the project.
Before now, phototaxis baffled microbiologists because of the small size of bacteria. Cyanobacteria are tiny, only a mere 0.0003 centimeters in diameter on average. When Mullineaux first learned that individual cyanobacteria cells could detect light, he was shocked: “I thought it was all completely impossible,” said Mullineaux. For many months, Mullineaux formulated new theories about the mechanism of phototaxis, but they were repeatedly proven wrong within the lab. Eventually, there was a breakthrough when Nils Schuergers, a graduate student working on the project, shone light from the side of a microscope at the cells. Small, focused spots of light appeared on the sides of the cells opposite from the light source. “As soon as we saw it, it was completely obvious,” said Mullineaux, “the cell must act as a lens.”
This new insight into how light travels through bacterial cells may be relevant to more than phototaxis. It raises a host of new questions about the amount of light cells are capable of absorbing. And since sunlight provides the energy for photosynthesis, a cell’s ability to absorb light controls how much carbon it can take in from the atmosphere.
