Some of the world’s deadliest diseases manifest when the body begins to betray itself. In cancer, mutated cells proliferate and overrun normal ones. Lupus, an autoimmune disease, occurs when the body’s immune system begins to attack its own cells. But what if the mechanisms of one disease could be used to counteract another?
This thought inspired recent work by James Hansen, a Yale professor of therapeutic radiology. Hansen transformed lupus autoantibodies — immune system proteins that fight normal proteins to cause lupus — into selective vehicles for drug delivery and cancer therapy.
His focus was 3E10, an autoantibody associated with lupus. Hansen and his team knew that 3E10 is able to penetrate into a cell’s nucleus to inhibit DNA repair and spark symptoms of disease. What remained a mystery was the exact mechanism by which 3E10 accomplishes nuclear penetration, and why the autoantibody is apparently selective for tumor cells. Unlockign these scientific secrets opened up new possibilities to counteract disease, namely, by protecting the very cells that the autoantibody might attack.
What Hansen’s team found was that 3E10’s ability to penetrate efficiently into a cell nucleus is dependent on the presence of DNA outside cell walls. When solutions absent of DNA were added to cells incubated with 3E10, no nuclear penetration occurred. With the addition of purified DNA to the cell solution, nuclear penetration by 3E10 was induced immediately. In fact, the addition of solutions that included DNA increased nuclear penetration by 100 percent. The researchers went on to show that the actions of 3E10 also rely on the ENT2 nucleoside transporter. Once bound to DNA outside of a cell, the autoantibody can be transported into the nucleus of any cell by the ENT2 nucleoside transporter.
“Now that we understand how [3E10] penetrates into the nucleus of live cells in a DNA dependent manner, we believe we have an explanation for the specific targeting of the antibody to damaged or malignant tissues where DNA is released by dying cells,” Hansen said.
This insight holds great meaning for cancer therapies. A lupus-causing agent, 3E10, naturally finds cancerous cells in the body because of DNA release. If this molecule were coupled with an anti-cancer drug, scientists would have a way of targeting that drug to tissue in need. It may seem counterintuitive, but what causes one disease could be harnessed to treat another.
The primary biological challenge for cancer therapy is to selectively target cancer cells while leaving healthy ones alone. The 3E10 autoantibody is a promising solution because it offers this specificity, a direct path to the tumor cells that will bypass all cells functioning normally. The molecule could carry therapeutic cargo, delivering anti-cancer drugs to unhealthy cells in live tissue.
The Yale researchers were pleased with their next step as well — they were able to engineer molecules that were tumor-specific. Mice injected with these molecules and then stained showed a presence of the antibody in tumor cells, but not normal ones.
Now, Hansen and his colleagues are looking into using the 3E10 and their engineered molecules to kill cancer cells. Since some cancer cells are already sensitive to DNA damage, inhibition of DNA-repair by 3E10 alone may be enough to kill the cell. Normal cells with intact DNA repair mechanisms would be likely to resist these effects, making 3E10 nontoxic to normal tissue. The researchers are working to optimize the binding affinity of 3E10 so that it can penetrate cells more efficiently and can exert a greater influence on DNA repair. The goal is to conduct a clinical trial within the next few years.
In the search for more effective drugs against cancer, answers can emerge from the most extraordinary places. “Our discovery that a lupus autoantibody can potentially be used as a weapon against cancer was completely unexpected. 3E10 and other lupus antibodies continue to surprise and impress us, and we are very optimistic about the future of this technology,” Hansen said.
The recent study was published in the journal Scientific Reports.
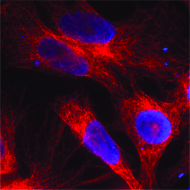
Cover Image: An immunofluorescent image of human colon cancer cells, stained red, penetrated by 3E10 scFv, stained blue. The nuclear-penetrating ability of 3E10 appears to selectively target cancer cells while leaving normal tissue unharmed. Image courtesy of Philip Noble and James Hansen.