Imagine you are a mother or father to be of a child who could potentially suffer from Down’s syndrome. Would you want to know as early as possible? Consider another scenario: imagine you could prevent the birth of a child suffering from cystic fibrosis, without having to deal with the ethical concerns of abortion – would you?
With the advent of Preimplantation Genetic Diagnosis (PGD), a reproductive technology that involves testing of embryos at the cleavage stage to select and transfer only the healthy ones, prospective parents now have greater control over these matters than ever before.
Previously, patients had the option of performing prenatal diagnosis, such as amniocentesis or chorionic villi sampling, to find out if their children were at risk for certain diseases. However, those methods can only be performed after women are already pregnant, leaving them with only two alternatives in the case of a positive test result: have a sick child or have an abortion.
PGD, on the other hand, “allows a patient to avoid the ethical dilemmas and potential medical complications associated with abortion,” says Dr. Maria Lalioti, Assistant Professor of Obstetrics and Gynecology at the Yale School of Medicine. This is because PGD is done before the implantation of the embryo – specifically, three days after fertilization.
“There is a window period of two days in which the embryos can be tested before they would have to be transferred back into the mother for implantation,” Lalioti added.
In addition to helping avoid the dilemmas of abortion, PGD also helps pregnant women avoid the dangers of spontaneous abortion of a diseased child. In most cases, the patients who opt for PDG are those who suffer from or carry a genetic disease or are aware that one is present in their family tree. Instead of learning the child’s genetic status in the middle of a pregnancy through prenatal diagnosis, an unaffected embryo could be implanted from the start.
Not for All: When Can PGD Be Used?
A typical pregnancy is not open to PGD. In order to be tested, the embryos must be accessible in an in vitro (that is, outside the body) environment.
Thus, only those pregnancies that result from in vitro fertilization (IVF) can use PGD. IVF is a form of assisted reproductive technology in which a sperm and an egg are mixed in a fluid medium outside the woman’s body. According to Lalioti, “although the IVF treatment is mainly catered to couples who are facing problems getting a child, about five percent of all IVF cases are done [solely] to make embryos available for PGD.”
Specifically, PGD may be used for patients whose embryos can be affected by single gene disorders such as thalassemia and cystic fibrosis or known chromosomal abnormalities, such as deletions, inversions and translocations. With these patients, PGD would conclusively detect and deselect embryos with the undesired mutations.
PGD may also be used to select embryos whose bone marrow matches that of an already sick child. The couples who desire PGD for this reason, says Lalioti, “have searched through the bone marrow registries, but to no avail. If they are still young and able to have another child, they may opt to receive the IVF treatment and test all the embryos for HLA matches with the first affected child.” This use of PGD is especially promising for diseases such as leukemia in which treatment relies on bone marrow transplants. Stem cells from the new child’s umbilical cord provide a potential reservoir for the transplant.



Does It Even Work? Problems of PGD
Before we discuss the ethical concerns associated with PGD, we must first look at practical ones.
PGD is currently offered to patients who are at an advanced reproductive age, have experienced repetitive IVF failure, or have suffered recurrent spontaneous abortion. The idea is that PGD could help women who are having trouble getting pregnant. Women at advanced reproductive age, for instance, are at high risk for producing embryos with aneuploidy, or an abnormal number of chromosomes.
In theory, PGD could help by detecting and selecting against these embryos. Embryos with aneuploidies, however, have a very low chance of implanting anyway; most will spontaneously abort before. Thus, some argue that it is not very compelling to conduct an invasive procedure for a problem that typically resolves itself naturally.
Dr. Pasquale Patrizio, Professor of Obstetrics and Gynecology, Clinical Practice Director, and Director of the Yale Fertility Center says, “By conducting PGD on women who are already advanced in maternal age and thus producing very few normal embryos, you might very well be arresting the growth of a potentially unaffected embryo.” Moreover, he adds, “With the current techniques we can only test for a limited number of chromosomes – specifically for nine out of the 23 pairs.”
As a result, according to Patrizio, the last year and a half has seen a drop in the number of requests for PGD.
Investigators also thought PGD might be able to help infertile couples who had undergone IVF treatment many times without success by transferring only the healthy embryos back into the mother. Although the logic behind this notion is sound, the success rate of IVF has not been shown to improve after PGD.
“Clearly, affected embryos are not the sole reason these women were not getting pregnant,” Lalioti explained. “We have failed to detect the problem with them; otherwise, we would not have resorted to PGD in the first place. Most of the times, these are cases of unexplained infertility.”
Techniques of Testing
Currently, there are three ways in which PGD can be performed. For single gene disorders and HLA typing, polymerase chain reaction (PCR) is conducted to amplify, or make many copies of, the gene of interest. After, the PCR products are digested with restriction enzymes, and these products are then sequenced or scanned for single nucleotide changes.
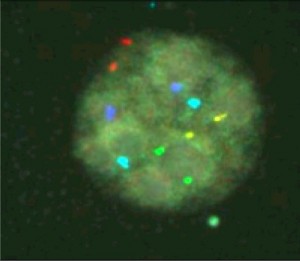
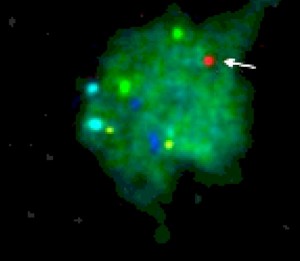
For the detection of larger copy number variations, or duplications and deletions in chromosomal regions, comparative genome hybridization (CGH) may be used. This technique compares subject DNA to normal reference DNA using array-based methods. Finally, to detect other large-scale changes, including aneuploidies and chromosomal rearrangements, fluorescence in situ hybridization (FISH) is used. This procedure utilizes several different colored probes to bind to various chromosomal regions and pick up abnormalities.
Before any of these techniques is carried out, the embryo produced via IVF is biopsied. After the egg is fertilized by a spermatozoon, it divides into the two-cell stage within the first day, the four-cell stage within the second day, and the eight-cell stage on the third day. It is in the eight-cell stage that the embryo is used for PGD; one of the eight blastomeres is removed for testing.
In order to conduct PGD by FISH, the selected blastomere is fixed and the first hybridization (binding of probes) is carried out for five chromosomes. Only if these chromosomes are normal will the second hybridization be performed to test the next four chromosomes. If all nine tested chromosomes are normal, the embryo will be transferred back into the mother for implantation.
CGH is a powerful tool because it is able to test the whole genome. However, this technique is extremely labor-intensive, and therefore cannot be performed in a timely manner. A successful technique must be capable of testing the embryo in the two-day window between the third day, when the eight-cell embryo is formed, and the fifth day, when the embryo has to be transferred back into the mother for implantation to occur. Unfortunately, more than two days are required to complete CGH, although according to Lalioti, new advances in the technology may enable more comprehensive and rapid whole genome screening.
Ethical Issues of Embryo Selection
PGD is not simply a scientific venture. It is also an ethical one. One controversial aspect, for example, is the use of the procedure to create an embryo as a bone marrow match for a sick sibling. Detractors argue that parents are creating a child not for his or her own sake, but to be used as a mere means for a previous child. Those in favor point out that the new child’s umbilical cord blood rich in stem cells will be used for the transplants, and taking these stem cells does not harm the child. Moreover, just because the new child is valuable for his cells does not mean parents will love or appreciate him, as a person, any less.
Another issue that PGD raises relates to sex selection and family balancing. “At the Yale Fertility Center, we perform PGD strictly for medical conditions. However, there is a delicate issue of whether or not to offer family balancing,” Patrizio said.
Patrizio, who also holds a Master in Bioethics (MBE), explained the distinction between sex selection and family balancing: “If a couple comes into the clinic and requests that their first and only child be of a certain gender, that is sex selection and the request is denied. For family balancing, you need to have at least two kids of one sex to be able to ask for a third and final child of the opposite sex.”
An initial concern “was a fear that the gender ratio would be skewed in favor of a particular gender,” explained Patrizio. But surprisingly, he noted, this is not the case, at least in the United States. Recent studies have shown that requests for the genders are on a 50:50 ratio.
Nevertheless, he added, “At the Yale Fertility Center, there are stringent criteria that have to be considered for family balancing. Sex selection is not offered in principle, unless there is a medical indication for it, such as a disease that is linked to sex.”
Some people may question the ethicality of PGD even when dealing with life-altering diseases. Should PGD be offered to a patient whose child has muscular dystrophy, for example? What is wrong with a baby born and confined to a wheelchair? One may argue that a baby in a wheelchair would not be able to live a good life – but who are we to decide what constitutes a good life?
Patrizio offered his take. “The key is to avoid suffering and to strive for the overall well-being of individuals. If a couple with limited resources, who will not be able to cope with a baby in need of constant care, comes in here hoping to give the gift of life to an unaffected baby for a specific, debilitating medical condition, why not? It is not my choice, it’s the couple’s decision.”
The Yale Fertility Center is hoping to collaborate with the Yale Stem Cell Center in the near future. With the permission of the parents, spare embryos that do not become implanted will be given to the Yale Stem Cell Center for potential research purposes. This collaboration hopes to aid in the development of cures for the very diseases that PGD selects against.
As Patrizio said, “Of course, if we knew how to cure cystic fibrosis today, PGD would be redundant. But the problem is that there is no cure.”
About the Author
EE-LYNN YAP is a sophomore in Morse College majoring in Chemistry. She is deeply interested in the ethical issues concerning reproductive technologies.
Acknowledgements
The author would like to thank Professors Pasquale Patrizio and Maria Lalioti for their assistance in preparing this article.



