
Consider the fruit fly –– it is tiny and, quite frankly, a nuisance to just about everyone. However, if you happen to be a biologist, the fruit fly might just be one of your favorite instruments. Drosophila melanogaster (as they are known taxonomically) are prime subjects not only for the trans-generational study of induced mutations but also for the study of insect ecology. As recent work by Yale Eugene Higgins Professor of Molecular, Cellular, and Developmental Biology John Carlson suggests, the expression of certain genes in Drosophila’s taste neurons indicates that taste is a more versatile tool in insects than had previously been anticipated.
While Drosophila has a variety of taste neurons whose expression of specific taste receptor genes is known, many neurons have not yet been matched to a taste receptor gene and have thus been labeled “orphan” taste neurons. Carlson’s team has discovered that a clade of 35 ionotropic receptor (Ir) genes is expressed not only in some of these “orphan” taste neurons but also in all of Drosophila’s taste organs. Unlike other Ir genes, this group, the IR20a clade, is not expressed in olfactory neurons. Accordingly, it also has a different function: determining whether the compounds that Drosophila’s taste organs touch are appetizing.
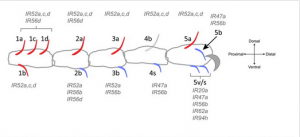
Furthermore, unlike those in humans, Drosophila’s taste organs serve functions not related to the consumption of food. The IR52c/d genes, for example, are expressed in sensilla located on the dorsal side of the leg where they are less likely to come into contact with food. The neurons in which they are expressed also show no increase in activity even when in contact with food. Rather, these sexually dimorphic genes are expressed more in males and located in the regions most likely to come into contact with a female during copulation.
Hypothesizing that the IR52c/d genes play a role in male sexual behavior, the researchers deleted them from a group of male Drosophila. They then measured how long it would take for half of the males in a regular, wild-type population to copulate, and compared this time to that of mutant males. The team observed a prolongation in the copulation time of the mutants, reinforcing the idea that the IR52c/d genes regulate the sexual behavior of male Drosophila.
With this new understanding of insect taste and the functionality of certain genes, it may become increasingly possible to tackle global issues caused by insects. By being able to “understand better how insects find the people they bite, how they find each other, and how they find the crops that they eat,” Carlson hopes that “we may be able to save a whole lot of people from disease and save a whole lot of crops from getting damaged.” Indeed, if a tactic that interfered with these receptor genes were developed, food could be made less palatable to insects or their reproduction could be hindered. Ironically enough, the fruit fly may hold all the answers to pest control.
