When you hear the word “medication,” what comes to mind? For the scientists in Dr. W. Mark Saltzman’s lab, the answer is not two tablespoons of Nyquil or even a Hepatitis B vaccine — it’s nanoparticles. In a recent study from the Yale University Biomedical Engineering Department, a team of researchers led by Rachel Fields and Elias Quijano created a new nanoparticle that can carry drugs capable of rewriting cell DNA. Made from biodegradable polymers, these tiny vesicles were designed specifically to transport synthetic polymers known as peptide nucleic acids (PNAs) to the cell nuclei, which can then permanently correct genetic mutations responsible for diseases like cystic fibrosis.
The best part is, it works.
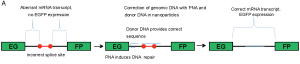
While gene therapy has been around for some 40 years, gene editing is a newer concept. Gene editing involves a “triplex-forming PNA,” which is a single stranded DNA-like sequence that can bind to a target mutation and then recruit DNA repair machinery to fix it. Collaborators of the Saltzman lab recently demonstrated that these PNAs could permanently reverse mutations in the gene that cause cystic fibrosis, a potential cure to a devastating disease. However, due to the nature of these peptides, the researchers needed a unique way to deliver the drug effectively and safely to the right parts of the body. To this end, Saltzman commissioned a nanoparticle delivery team. Because of their customizability, nanoparticles can make for excellent drug delivery systems when designed with the right chemistry. For Elias Quijano and the rest of the team, the biggest challenge was cellular uptake. “For our particles, targeting really wasn’t the issue,” Elias explained. “Instead we were concerned with the local delivery and uptake across the entire lung, in hopes of achieving editing throughout lung cells.”
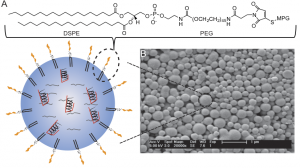
Elias and his colleagues focused on optimizing the composition of the surface of their nanoparticles to improve the permeability of the drug across membranes. They developed nanoparticles from a synthesis of two biodegradable polymers and then decorated the surface with short peptides that promote nanoparticle localization to the nucleus. The researchers then tested the efficacy of these nanoparticles by injecting them into cells that would fluoresce if properly repaired. They found that their modified particles successfully localized to the pulmonary tissue in mice and that the efficiency of their particle uptake was 80 percent greater than that of untreated nanoparticles.
“This success is only a stepping stone,” Elias explained. “As we speak, our collaborators are working on improving this technology to create a robust solution for real patients with cystic fibrosis.”
It is this interdisciplinary environment at Yale that has made Elias even more committed to his work and also excited to move forward. “There are always scientists contributing to new innovations, working together to advance the biomedical engineering world,” he pointed out. In the future, the team hopes that nanoparticles will be used to treat cystic fibrosis in people, but until then they will keep designing better polymers and building better vehicles.
