Microglia are generally thought of simply as the innate immune cells of the brain, responsible for removing dead neurons via phagocytosis. However, Ana Badimon and her colleagues discovered that microglia play a larger role in the human body. While a member of professor Anne Schaefer’s lab at the Icahn School of Medicine at Mount Sinai, Badimon et al. discovered that microglia are also neuromodulators—they control neuronal activity.
Other studies have found that alterations in microglial function, resulting in problems in neuronal activity and neuronal connectivity, have been linked to autism, psychiatric diseases and neurodegenerative diseases, all of which are characterized by changes in neuronal activity.
Badimon and colleagues started experimenting by removing microglia from mice brains. “When we activated neurons pharmacologically in the absence of microglia, we found that this resulted in an exaggerated neuronal response characterized by hyperactivity and seizures,” Badimon said. For Badimon and colleagues, this observation suggested that microglia are necessary to reduce excessive neuronal activity in order to maintain proper brain function.
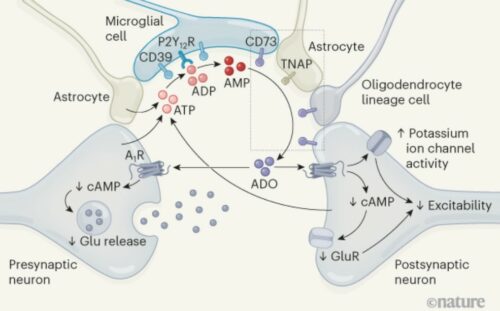
Badimon and her team then sought to explain the mechanism of microglia-driven inhibition of neuronal activity. Increased neuronal activity is often concomitant with higher levels of synaptic adenosine triphosphate (ATP), which is released locally by neurons or astrocytes. “This could be like a global signal that when there’s too much neuronal activity, then there’s increased ATP release,” Badimon said.
With the help of collaborators who performed live imaging, the researchers found that ATP serves as a signal for microglia, attracting them to active neuronal synapses. By pharmacologically blocking the microglia’s ATP-sensing P2Y12 receptor, microglia no longer preferentially contact the active synapses.
In addition to acting as a chemoattractant for microglia, ATP can be cleaved into adenosine, which then acts as a neuromodulator to dampen neuronal activity. This is a two-step process: CD39 enzyme cleaves ATP into AMP and then CD73 cleaves AMP into adenosine. In the brain, CD39 is primarily expressed by microglia. Badimon et al. showed that genetic ablation of CD39 on microglia induces neuronal hyperresponsiveness and seizure behavior, thereby demonstrating the essential role CD39 plays in generating adenosine to modulate neuronal activity.
Finally, Badimon and coworkers showed that this feedback control by microglia is likely a homeostatic mechanism required to maintain brain health. Recent studies have shown that in some diseases, such as Alzheimer’s and Huntington’s disease, P2Y12 and CD39 expression is downregulated in activated microglia. This results in an inability of microglia to sense synaptic ATP activity and consequently a loss of microglia-mediated neuronal modulation, which may underlie some of the phenotypes seen in these diseases.
These findings suggest that microglia may change neuronal behavior in response to a peripheral infection or perturbation. Furthermore, restoration of normal microglial function may serve as a potential therapeutic for some neurodegenerative diseases in the future. These findings raise the possibility that microglia may also be able to sense other molecules or proteins released by neurons or astrocytes and induce a response mechanism that has not been identified yet.
“Neurons were always the main focus in neuroscience,” Badimon said. “Now scientists are investigating the interplay between the different cell types in the brain, and it is bringing to light the important role that all the cells play in keeping the brain healthy and functional.”