Every society needs a group of superheroes. And as a society of proteins, organic molecules, and nucleic acids, cells are no different. To defend against pathogens, certain proteins within the cell work vigilantly to secure its safety. One group of these vigilantes hails from a set of mysterious genes termed the apolipoprotein L (APOL) family. Twenty years ago, the discovery of APOL1, which functions outside of the cell to defend against extracellular parasites, led scientists to believe that the other five APOL genes may defend against intracellular pathogens since they lack a secretion signal. Led by postdoctoral fellow Ryan Gaudet, the MacMicking lab at Yale and collaborators unearthed the function of one of these five genes, APOL3, which codes for a protein that attacks Gram-negative bacteria such as Salmonella.
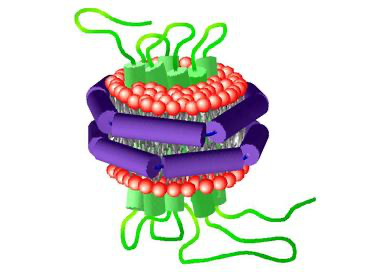
APOL3 interacts with another host defense protein, guanylate binding protein (GBP1), which autonomously binds to the sugar-rich surroundings of Gram-negative bacteria. When GBP1 invites APOL3 to the inner membrane of Gram-negative bacteria, APOL3 kills the pathogen by dissolving the lipid membrane, essentially ripping apart the bacterial membrane in the cytosol.
With great power comes great responsibility: APOL3 needs to discriminate between self and non-self-membranes. APOL3 doesn’t surround lipids within the cytosol without specificity—that would be dangerous. A key ingredient in host cell membranes, cholesterol, is an inhibitor to APOL3 that prevents self-destruction. “Cholesterol makes APOL3 less able to insert its hydrophobic component into the membrane,” Gaudet said.
Gaudet is optimistic about the many avenues of exploration for APOL3, including future work investigating the protein’s role in vivo.