Image courtesy of Tyler Greer.
For Jayme Locke, the hardest part of being a transplant surgeon is knowing that a gold standard treatment exists, yet not being able to use it. In the face of end stage kidney disease, the biggest barrier to treatment is the ongoing organ shortage. With demand drastically exceeding supply, a radical solution is imperative. That solution oinks, rolls in mud, and can play videogames with their snouts. Enter the pig: an innovative solution to the organ supply crisis.
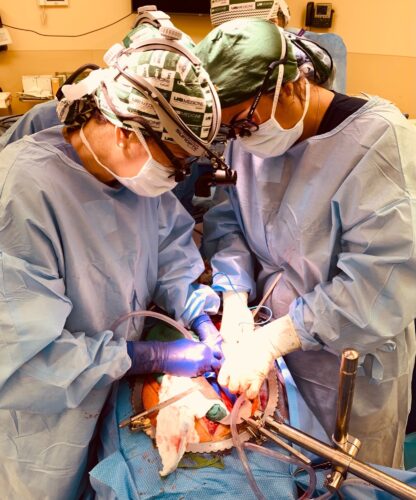
At the University of Alabama at Birmingham (UAB), Locke was the lead surgeon in a study that performed the world’s first transplant of genetically modified kidneys from a pig into a human. According to Locke, there are eight-hundred thousand Americans with kidney failure, and within that group, six-hundred thousand are on dialysis. Only around ten percent of these Americans make it to the kidney transplant waiting list, and a measly three percent receive kidney transplants each year.
“We know kidney transplantation is the cure for kidney failure. We want to be able to offer the cure to everyone in need,” Locke said.
Only about thirty-five percent of people survive past eight years on dialysis. Meanwhile, a kidney transplant offers a success rate of ninety-five percent (for deceased donor transplants) to ninety-eight percent (for living donor transplants). A kidney transplant also improves a person’s quality of life. Kidney failure is an end stage disease—if it is not fixed, the patient will die. Therefore, the prospect of having an organ on the shelf, waiting for anyone who needs it, is truly revolutionary.
In their search for a donor source animal, pigs stood out. In order to meet the current and projected demand for kidney transplants, the research team needed an animal that could rapidly reproduce large litters. “The domestic pig was chosen because of its ability to ‘scale-up.’ They also have a lifespan close to thirty years, which is great when it comes to kidney longevity,” Locke said.
However, since the pig is still relatively foreign to the human immune system, it was crucial to edit ten genes to make the pig kidney more “human.” These edits, along with the standard immunosuppression involved in human-to-human transplantation, allowed the human body to tolerate the pig kidney, and for the pig kidney to sustain the person.
The specific genetic modifications included the targeted insertion of two human complement inhibitor genes, two human anticoagulant genes, and two immunomodulatory genes, in addition to the deletion of three pig carbohydrate antigens and the pig growth hormone receptor gene. The end result was a herd of genetically engineered pigs whose inability to express red blood cell antigens allows them to serve as universal donors.
According to Locke, one of the greatest challenges in xenotransplantation is understanding tissue compatibility between the porcine donor and human recipient. If the tissues do not match between the donor and the recipient, the latter will reject the organ within minutes of establishing blood flow. To overcome this challenge, the team reached out to Vera Hauptfeld-Dolejsek and Julie Houp, co-directors of the UAB Histocompatibility Lab, who developed a novel assay specific to pig-to-human transplant.
For the assay, the pig donor’s red blood cells were combined with the human recipient’s serum in a crossmatch. This assay tested whether a kidney from a pig could tolerate an adult human environment. The negative control was pooled human male AB serum, while the positive control serum contained IgG, an antibody known to react with porcine cells. In the study, the human recipient’s blood was mixed with pig cells to demonstrate a negative crossmatch, allowing the transplantation to proceed. This ability to predict compatibility between the pig xenograft and the human recipient would prove to be very accurate.
The second hurdle of this study was testing for hyperacute rejection without harming a living person. Hyperacute rejection occurs a few minutes after the transplant as a result of the antigens being completely unmatched—the body’s immune system treats the transplanted organ as a foreign object and attacks it. The team’s solution was to create the first human preclinical model.
The Parsons model was named in honor of Jim Parsons, a fifty-seven-year-old man from Huntsville, Alabama. Parsons had been a registered organ donor through Legacy of Hope, which is Alabama’s organ procurement organization. In light of his sense of adventure and desire to make a difference, the Parsons family sought to pay tribute to his character. After Parsons was declared brain dead and his organs were deemed unsuitable for donation, the Parsons family ultimately consented to him serving as the first preclinical model for this groundbreaking study.
While human brain death had already been used to harvest organs for human transplantation, it was novel to leverage brain death as a preclinical human model. One critical concern to be tested via the model was the vascular integrity of pig kidneys. Pigs do not have the same mean arterial pressure as an adult human being, so whether the transplanted kidney would be able to hold its integrity was unknown. Another goal of this preclinical model was to determine whether the genetic engineering, coupled with a negative prospective crossmatch, were sufficient to prevent hyperacute rejection.
During surgery, the two pig kidneys were positioned in the exact anatomic locations used for human donor kidney transplantation, and employed the same attachments to the renal artery, renal vein, and the ureter.
“In the present study, the crossmatch was performed prior to transplant—just as happens in human-to-human transplantation—and it was negative, predicting there would NOT be hyperacute rejection. The only way to validate this was to perform the actual transplant and demonstrate the kidney turned pink and made urine. We did this leveraging the Parsons Model, and in so doing, answered key safety questions without risking the life of a living person,” Locke said.
To the team’s delight, the pig kidneys reperfused promptly in the same manner as human transplants. The kidneys retained optimal color and turgor, the vascular connections between donor organ and recipient stayed intact, and there were no major bleeding episodes. Within around twenty minutes, the right kidney started making urine, later followed by the left. The ureter had successfully carried urine from the pig kidney into the human bladder. There was no sign of hyperacute rejection.
This success proved the accuracy of their crossmatch and firmly established brain death as a viable preclinical model for studying the human condition—where a treatment’s safety and feasibility may be tested without doing harm to someone. Such a model would extend far beyond xenotransplantation—there are many diseases that have yet to be understood, along with new techniques and devices in need of testing before use on a living person.
In a pathogen-free facility, a herd of pigs awaits. These pigs will be the proper size for adult human transplantation by June 2022. The team hopes that the FDA will approve their Investigational New Drug Application, and thereby allow the launch of a phase I clinical trial in living persons, a process Locke is hopeful to begin in 2022.
Particularly in the era of COVID-19, regulatory agencies will rigorously assess the transmission of viral diseases from pigs to humans. In this study, the team tested the pig pre-procurement to ensure that the pig did not have any diseases. Further, the human recipient’s blood was tested post-transplantation to prove the absence of pig-derived infections or diseases.
Locke is hopeful that, as early as five to ten years from now, the pig xenograft kidney will be available for widespread use. She envisions xenotransplantation and allotransplantation as complementary; together, there is the real potential to completely eliminate the waiting list and wipe out the organ shortage.
For now, our ability to pee may be secured, one pig at a time. Who knows what organ or animal will be next.