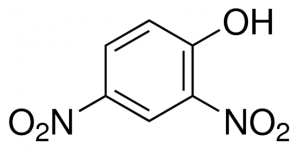
The molecule behind a weight-loss pill banned in 1938 is making a comeback. Professor Gerald Shulman and his research team have made strides to reintroduce 2,4 dinitrophenol (DNP), a once toxic weight-loss molecule, as a potential new treatment for type 2 diabetes.
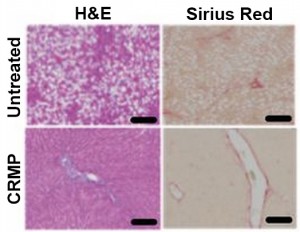
Patients with type 2 diabetes are insulin resistant — they continue to produce insulin naturally, but their cells cannot respond to it. Previous research by the Shulman group revealed that fat accumulation in liver cells can induce insulin resistance, non-alcoholic fatty liver disease, and ultimately diabetes. Shulman’s team identified DNP, which reduces liver fat content, as a possible fix.
DNP was banned because it was causing deadly spikes in body temperature due to mitochondrial uncoupling. This means that the energy in glucose usually harnessed to produce ATP is instead released as heat. Shulman’s new study offers a solution: CRMP, a controlled release system that limits the backlash of DNP on the body. CRMP is an orally administered bead of DNP coated with polymers that promote the slow-release of DNP, which prevents overheating.
So far, findings have been promising: no toxic effects have been observed in rats with doses up to 100 times greater than the lethal dose of DNP. “When giving CRMP, you can’t even pick up a change in temperature,” Shulman said.
Results also include a decrease in liver fat content by 65 percent in rats and a reversal of insulin resistance.
“Given that a third of Americans are projected to be diabetic by 2050, we are greatly in need of agents such as this to reverse diabetes and its downstream sequelae,” said Rachel Perry, lead author of the study.
Cover Image: Rachel Perry, the study’s lead researcher, measures out 2,4-dinitrophenol (DNP) for experimentation. Dr. Perry is a postdoctoral fellow in the Shulman Lab. Image courtesy of Cheryl Mai.

