Every year, more than 200 million cases of malaria result in nearly 700,000 deaths. Most of these fatalities occur in children under 5 years of age in Sub-Saharan Africa. Although malaria has been curable since the mid-1800s, this treatment involves an intensive three-day regimen of various drugs, which is not always effective and is difficult for some patients to complete in full. However, a research team at the University of Cape Town plans to combat malaria with a new single-dose cure.
This single-dose drug candidate, developed by chemistry professor Kelly Chibale and his team, can kill malaria parasites at most stages of their life cycle. The drug produced no side effects in animal trials. In contrast, current methods of treating the mosquito-borne illness cause many adverse side effects and are limited in their efficacy. Chibale’s potential single-dose cure, code-named MMV390048, has entered Phase 1 clinical trials in South Africa to test optimal dosage. If trials proceed as Chibale hopes, MMV390048 will have tremendous public health implications in low-income African countries, greatly reducing the burden of malaria in these regions.
The onset of malaria is marked by flu-like symptoms, followed by high fever, coma, and potentially death. Malaria is caused by parasitic unicellular protozoans, which are transmitted by mosquitos. The protozoans attack and reproduce in the human body, first residing in the liver and then reproducing in red blood cells. This multi-step life cycle was a major hurdle for earlier treatments, although the new drug candidate addresses this obstacle. Quinine was the first medication used against malaria. Scientists believe quinine facilitates the aggregation of cytotoxic heme molecules within malaria parasites, which ends up killing them. However, quinine causes significant adverse side effects, including tinnitus, blurred vision, and confusion. It is only effective when the parasite is in the red blood cell phase of its life cycle. Malaria parasites also easily develop resistance to quinine.
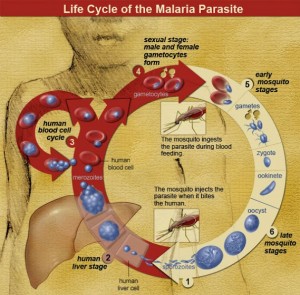
Additional anti-malarial drugs have since been developed, including arteminisins, amodiaquine, and sulfadoxine. Since malaria parasites have become resistant to the vast majority of these drugs independently, doctors must administer them together in combination therapies in order to adequately cure the disease. Since these drugs only inhibit certain stages of the parasite’s life cycle, doctors must administer them over a course of three days to completely rid the body of malaria. The full-course three-day regimen is not feasible for patients who cannot afford the cost of hospital visits or who have low compliance for medical treatment, which is true of a large share of patients, especially when suffering from fever and nausea.
MMV390048 presents a solution to most of these issues. Scientists at Griffith University in Australia first discovered an MMV390048 precursor in 2012 as a part of a potential new class of antimalarial drugs. These scientists passed on their research to Professor Chibale, who worked to optimize this compound and to further explore its antimalarial potential.
In 18 months, Professor Chibale and his team perfected a suitable drug candidate for preclinical development. This process usually takes 1-5 years. Chibale’s team found that the drug worked successfully in animal trials, killing malaria parasites at most stages of their life cycle. They hypothesize that MMV390048 acts as a kinase inhibitor. Kinase reactions, which transfer high-energy groups from one molecule to another, are critical in cells, as they speed up vital metabolic pathways. By inhibiting kinase reactions specifically in protozoans, MMV390048 is highly toxic to these parasites at any stage in their life cycle.
In addition to its scientific merits, MMV390048 is unique because of its African origin. “This is the first-ever clinical molecule that’s been discovered out of Africa, by Africans, from a modern pharmaceutical industry drug discovery program,” said Chibale in an interview with National Geographic. Medicines for Malaria Venture, the Swiss non-profit product development partnership that first facilitated collaboration between the group at Griffith University and Chibale’s team at the University of Cape Town, expresses hope that this finding will be one of many South African scientific accomplishments to come.
A single-dose cure for malaria has sparked hope for a more streamlined approach to treating this disease that causes a tragic number of deaths every year. If clinical trials in humans are successful, MMV390048 will not only be an accomplishment for Africa, but also for the global scientific community.
