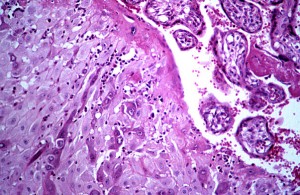
Pain and swelling for months. Crushing of internal organs. Immunosuppression. These sound like the symptoms of a debilitating disease or serious injury. Yet they are the signs of a natural condition which about 100 million people around the world have at any given time – placental pregnancy. As placental mammals ourselves, we often do not realize that this trait is a very radical phenotype. Professor of ecology and evolutionary biology Günter Wagner and colleagues have conducted genomic analysis of cells from an array of pregnant mammals to uncover how this dramatic development arose.
In a February 2015 article in Cell Reports, Wagner’s team published the results of a five-year project which examined the transcriptomes of decidual cells of a wide range of animals. Transcriptomes are RNA copies of DNA which are later translated into proteins, and decidual cells are specialized uterine cells which the mother uses to communicate with and feed the fetus during pregnancy. Wagner found that transposable elements, repetitive DNA sequences that can move themselves into different areas of the genome, are the likely vehicles for the radical genome-wide changes that led to the evolution of placental pregnancy.
Most animals, from fishes to frogs, lay eggs. The oldest mammals (monotremes such as echidnas) still lay eggs, and the second-oldest (marsupials such as kangaroos) produce tiny underdeveloped young which mature in the mother’s pouch. Only the youngest group of mammals, plancental mammals (eutherians) bear fully-grown offspring that develop as invasive embryos within the mother.
The traditional view on evolution is that novel traits arise gradually via base pair substitutions or duplications. However, Wagner’s group was skeptical that small changes like these could have achieved such a dramatic change from egg-laying or the production of immature young to full-blown placental pregnancy. To uncover an alternate solution to this puzzle, they performed genome-wide scans of decidual cells from animal samples of various species.
By using biochemical methods to detect which parts of the decidual cell genomes were actively transcribed or regulated, Wagner’s group discovered that transposable elements were key players in the production of the decidual cell phenotype.
Wagner notes that the nature of transposable elements makes them viable agents for evolutionary change. “Since transposable elements came from retroviruses, they already contain gene regulatory components, so just by moving close to other genes, they have the potential to cause massive regulatory alterations in a cell,” said Wagner.
The group also found that many of the uterine regulatory elements are much older than pregnancy itself, supporting the theory that these regulatory elements arose from transposable elements that were already present in the cell before assuming pregnancy-related functions.
Drawing conclusions from the project’s enormous dataset required the input of scientists from a wide range of disciplines. “These days, large joint efforts are the key to many biology studies which include both experimental genomic and comparative aspects,” said Wagner. “In addition to the people in my lab, we needed the people who provided the specimens, people who are experts in transposable elements, and people who already had data on decidual cells in mice and humans.”
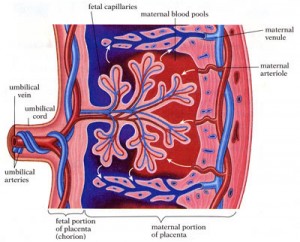
Now that they have achieved a broad genomic view of the patterns of genetic changes which led to placental pregnancy in mammals, Wagner and his colleagues plan to conduct more focused studies on the decidual cell core gene regulatory network. In January 2015, the Wagner lab was awarded a million-dollar grant from the John Templeton Foundation to study how this network functions and how it changed to give rise to decidual cells.
However, Wagner’s findings have implications which extend beyond the field of pregnancy research into all studies involving genomics. “A lot of traditional genomic analysis excludes repetitive elements such as transposable elements because they’re a pain in the neck to deal with,” said Wagner, pointing out that fifty percent of the human genome is made up of such repetitive elements. “Yet our findings suggest that they are actually really important, so we can’t afford to ignore them anymore.”
Cover Image: Professor of ecology and evolutionary biology Günter Wagner, far left, with the members of his Yale lab. Image courtesy of Günter Wagner.
