What comes before baby steps?
Designer babies, genetic remodeling, risks in clinical trials: in today’s rapidly evolving field of biomedical engineering, ethical dilemmas rage. But for Rachael Putman (TC ’19), it’s simple: “The whole point of medicine is to help people heal. If we can take gene editing and make it do that in a safe and effective way, then it’s something that we should pursue,” she said.
Putman, a Missouri native, matriculated to Yale College without any prior research experience, but was quickly hooked by Professor Mark Saltzman’s master class on in-utero brain research. She reached out to Saltzman for a position in his lab, and took her first steps in research. After honing her technical laboratory and research skills under Saltzman’s tutelage, Putman began investigating in vivo gene editing in mouse fetuses alongside Adele Ricciardi, an MD-PhD student at the Yale School of Medicine.
As a collaborative effort between the Saltzman laboratory and the laboratory of Professor Peter Glazer, Putman and Ricciardi are testing the safety and efficacy of triplex gene editing technology, which uses peptide nucleic acids (PNA). Unlike other gene editing techniques, such as CRISPR and TALEN, PNAs allow for a safer gene repair by avoiding double-strand breaks in the host DNA. Once the PNA is safely inserted into the cell, it binds to the targeted mutation in the DNA and creates a triple strand. The cell then recognizes and removes the PNA-bound segment from the genome, creating a gap. A short piece of DNA delivered with the corrected base pairs is then used as a template to fix the gap created by PNA.
“We tried to focus on safety and to see if we were getting any off-target effects,” Putman said. “Through our analysis, we couldn’t find any, which may give it an advantage over other technologies like CRISPR.”
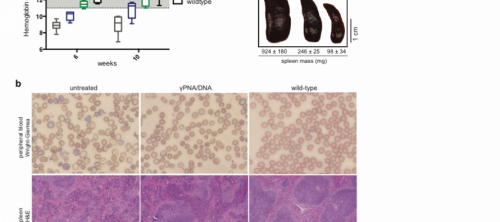
Putman and Ricciardi apply this gene editing pathway to cure human models of disease, such as cystic fibrosis, β-thalassemia, and sickle cell, in mouse fetuses. Nanoparticles that contain PNA strands are delivered to mouse fetuses and the mice are tested for the absence of disease after birth. “We were able to show that we got disease improvement after birth; the mice were healthy when they were born,” Ricciardi said.
A successful direction in this project led Putman to an even more fundamental question: can we do this even earlier? In other words, if site-specific genome editing works in fetuses, could it work in single-cell embryos? In many genetic diseases, organ damage can take place early on during the fetal stage of development. However, these diseases can often be identified at the embryonic stage if a mother undergoing in vitro fertilization chooses to screen for genetic diseases before implantation. If gene editing can be performed within the first days of life, there is a possibility that the fetus will undergo completely healthy organ development. With hopes of clinically translating her work with mice embryos, Putman embarked on her own project: Synthetic Nanoparticle Delivery to Mouse Embryos for Site-specific Genome Editing.
For her work with these two projects, Putman received the Arnold and Mabel Beckman Foundation award in 2017 and presented her work at the Beckman Scholars Annual Research Symposium for the following two summers in Irvine, CA. Putman was one of two Yale College students in the class of 2019 to receive this distinction.
Outside her research, Putman is the president of Demos, a service group that makes science education more accessible to students in New Haven public elementary schools. She also teaches a class about genetics to neighboring middle and high school students through Splash at Yale. As Putman serves younger scientists through her volunteer work, she attributes her own successes to mentorship. “I had two really wonderful teachers throughout middle school and high school,” Putman said. “What they taught me was that we all have things we’re good at and are passionate about. What is more important is to follow your passions and see where they lead you.”